The factors that determine whether a protein is druggable are complex. The question is whether it is possible to find an inhibitor that will be able to conform to the requirements of the protein’s location, shape and lipophilicity.
The first challenge to consider is the target location. This determines the properties required of the compound for it to reach the location at sufficient concentration for inhibition. This has to be balanced against the specific features required of the compound to fit the binding site within the protein in order to achieve inhibition.
Sometimes, these two parameters are diametrically opposed to each other and thus the target is not easily druggable. However, in many cases innovative solutions can often be found once there is wide enough understanding of the specific system.
The serine proteases are a case in point. Many of them require a highly basic arginine-like moiety such as benzamidine to access a key binding site location: the S1 pocket. This same benzamidine moiety disallows penetration through membranes. One solution for this, rather than defeat, is a pro-drug – a membrane soluble benzamidine analogue such as etixilate.
A more informative gauge of drugability of a target is the extent of the requirement for lipophilicity. This is the bane of medicinal chemistry, as almost inevitably lipophilicity increases during lead expansion (as presented by Mike Hann at CDDD Verona). Getting the drug to the target rather than to lipid membrane compartments and other targets is very difficult. Off target effects and toxicity both increase with lipophilicity. Lipid binding GPCRs such as prostaglandins and other receptors such as the cholesterol binding / transporter proteins are very challenging drug targets.
Cresset consultants work across a range of protein targets, as illustrated below. The chart classifies Cresset’s recent consulting projects according to target class. Proteases and protein-protein interactions are the most challenging targets.
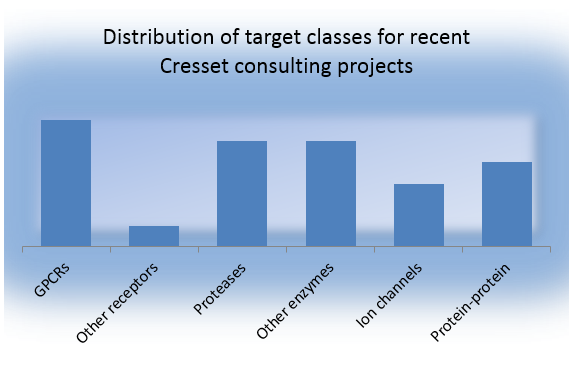
Figure 1: The distribution of target classes for recent Cresset consulting projects.
Receptors
Receptors are found inside cells or on cell membranes. When activated by a ligand, the receptor performs a specific biochemical action.
Receptors can act as an intermediary between a ligand and a protein across a cell membrane. The ligand activates the receptor so that it can activate the protein, forming a ligand-receptor-protein complex, as in the case of GPCRs.
G-protein coupled receptors (GPCRs) are one of the most important receptor families of proteins. About 40% of all available drugs act on GPCRs.
Conversely, nuclear hormone receptors act inside the nucleus of the cell and are involved in the regulation of gene expression which in turn is affected by small molecule hormones such as steroids and synthetic molecules such as Bis phenol A (BPA) – an endocrine disruptor leaching from polycarbonate containers.
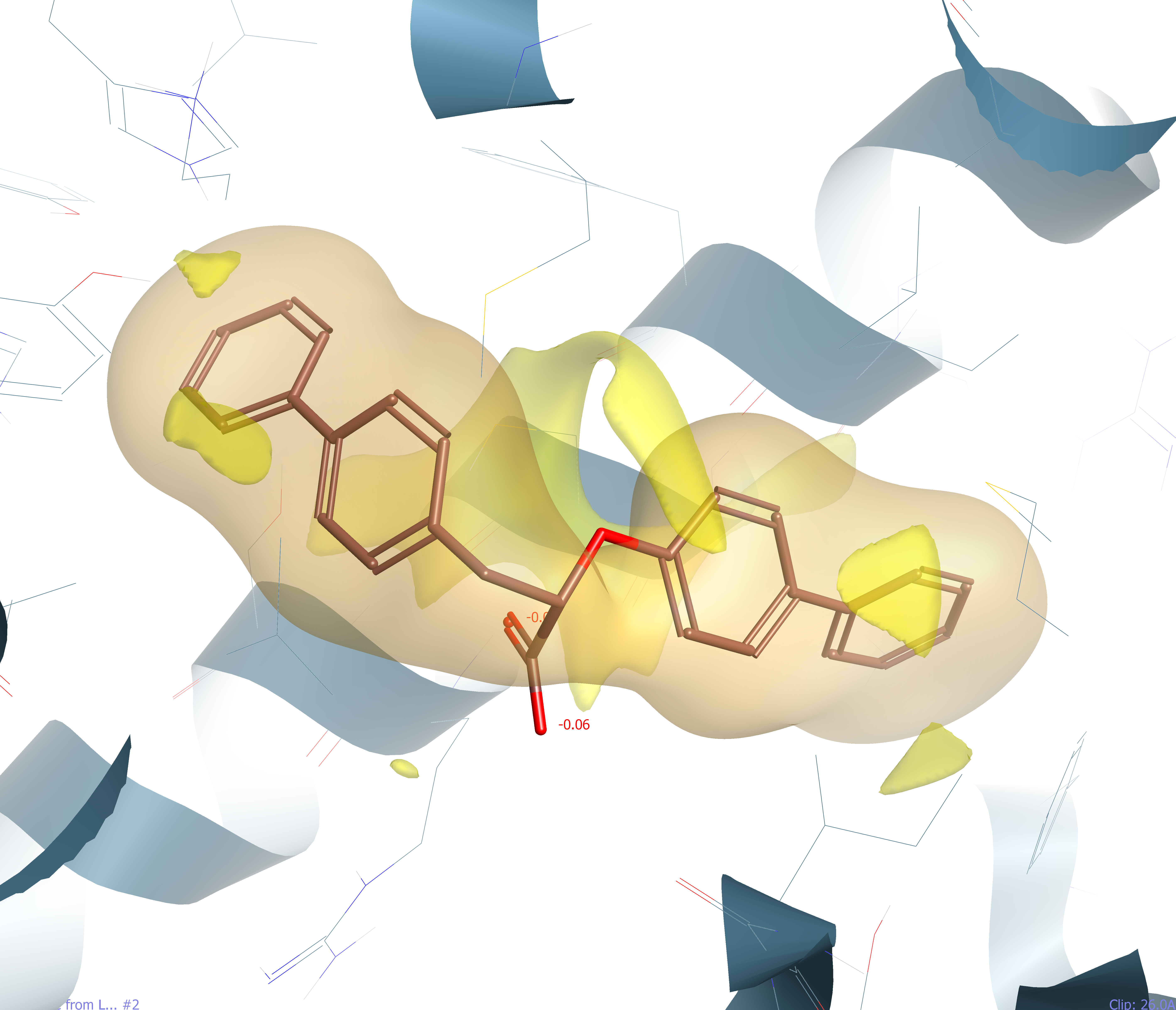
Figure 2: A highly lipophilic agonist ligand (slogP 6.1) of the PPARgamma nucleur hormone receptor PDB:4JL4 showing hydrophobic surface (brown) and vdW interaction hotspots (in yellow).
Enzymes and proteases
Enzyme inhibitors are an important group of drugs. They are effective in therapeutic areas as diverse as cancer, depression, cardiovascular disease and pain.
Protease enzymes break down proteins and peptides. AIDS is treated with HIV protease inhibitors. Over 700 human proteases have been identified so far. However, it is notoriously difficult to develop drugs for protease targets due to selectivity issues and property space.
Ion channels
Ion channels control the flow of ions such as sodium, calcium or potassium across a cell membrane. They play an important role in regulating many physiological processes, so can be very useful targets.
Protein-protein targets
Protein-protein interactions are a medial step in many biological processes. Designing a compound that will inhibit these interactions is a promising way to intercept mechanisms that have an adverse effect on a disease condition.
The challenges of designing suitable inhibitors relates mainly to the large surface area of the proteins. The computing power required to model the proteins means that identifying suitable binding sites can be a slow process.
However, there is a high therapeutic potential in designing suitable small molecules for protein-protein targets.
Working towards innovative solutions
The type of protein target has a fundamental impact on the nature of a drug discovery project.
Cresset’s computational methods are not dependent on knowledge of the target – we can take a ligand based approach to drug design. However, if there is structural information available for the target then we use it.
Cresset consultants are always keen to take on new challenges. Contact us to discuss how we can work with you to arrive at innovative solutions.

