Cresset User Group Meeting 2024
Register to join us for our upcoming User Group Meeting REGISTER HEREWe’re delighted to have colleagues from across industry and ...
News
There has been a revival of interest in covalent inhibitors in recent years. This has culminated in a growing list of diverse proteins that have been successfully targeted, that are outside the well-trodden protease area.1-4
An older non-protease example is Clopidogrel. This P2Y12 inhibitor requires activation in vivo (through oxidative metabolism) to generate the electrophile intermediate (sulfenic acid) that forms a covalent disulphide bridge to a cysteine residue in the target GPCR. Unravelling the true mechanism of this compound led to second generation inhibitors which further exploit the covalent mode of action5.
One more recent, and well documented, non-protease example is in the field of oncology, with the drug Ibrutinib. The covalent binding of the inhibitor to the target kinase BTK demonstrates multiple benefits:
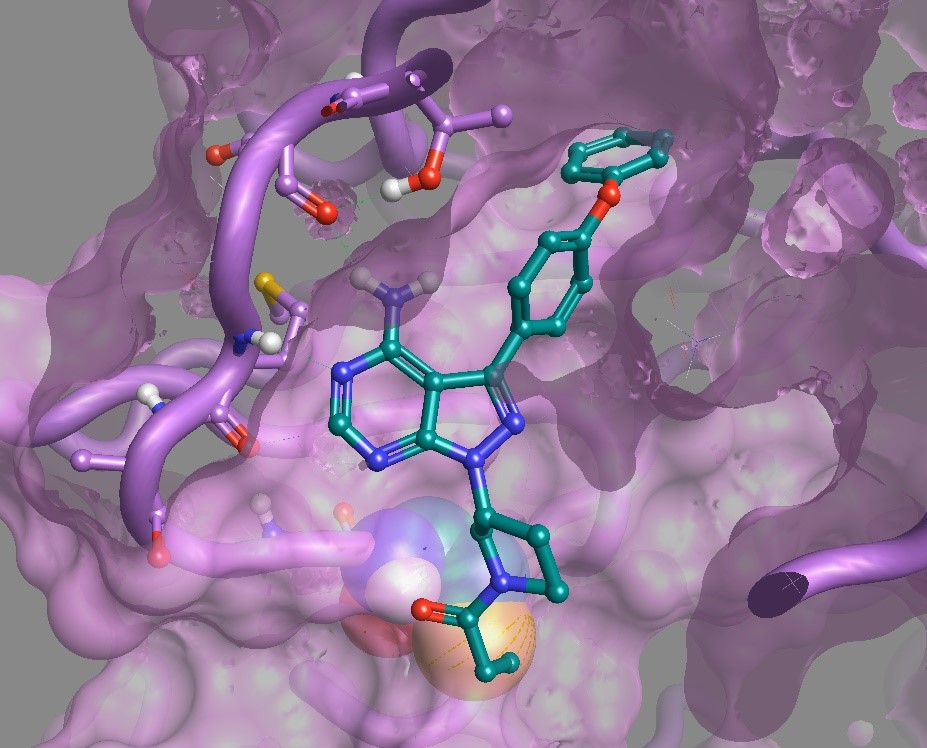
From a safety point of view, a method of achieving high efficiency/potency of any drug should translate into a lower dose that is ideally below any potential toxicity threshold (very few drugs are given at =< 10mg per day!). Similarly, prolonged duration of action, a consequence of the covalent inhibition, can result in less frequent dosage regimes that could be very beneficial – depending on the exact pharmacology.
Mainly, because this is not a panacea – some proteins are more quickly processed, turned over or are mutated. The BTK case itself is an unfortunate example of the latter; the resistance mutation C481S produces a protein that cannot undergo the reaction with the inhibitor and ultimately limits its utility in patients. What may have more generally hampered covalent inhibition strategies in the past, observed particularly in the field of protease inhibitor design, was the high electrophilicity and poor selectivity. The very justifiable fear of off-target activity and threat of unresolvable toxicity issues, has contributed to the adoption of a dogma which still significantly disfavors the approach across the industry. The lack of a wider uptake of the approach since then has been exacerbated, perhaps, by the industrialization of experimental design and/or the lack of attention to the kinetic behavior of these systems. One size doesn’t really fit all!
An appreciation of the kinetics of molecular recognition and its impact on drug efficacy (Figure 2) has had a resurgence; together with the introduction of biophysical techniques, such as Surface Plasmon Resonance (SPR), which allow the routine resolution of otherwise painfully elusive kinetic parameters such as kon/koff.
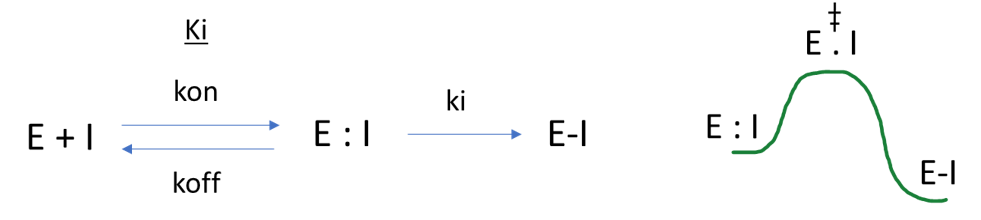
The observed rate of inhibition for a covalent inhibitor is effectively dependent on the ratio of the rate constant for the chemical transformation of the inhibitor (ki), relative to the rate of unbinding (koff). We can introduce a level of catalysis in this system if we design appropriate geometries that lower the energy of (i.e. favor) the E : I complex7. If we are talking about hand-crafting optimum molecular geometries, then we are usually thinking about utilising computational chemistry as a technique.
Advances in the covalent inhibition area have clearly arisen from a systematic investigation into the modulation of the intrinsic reactivity of these electrophilic ligands. Covalent inhibitor design, in its more current form, now reduces reliance on the Ki, and drives optimization through binding (i.e., reducing koff) rather than reactivity. This more thoughtful approach is an essential step towards maximizing selectivity and minimizing off target activity. In parallel with the ‘much needed’ experimental investigations, a range of computational techniques are required for modern covalent inhibitor design strategies. These include:
We anticipate that this approach will prove increasingly useful, particularly for otherwise intractable biological targets involving large and complex protein-protein contacts.
Cresset Discovery Services’ experienced scientists work alongside your chemists to solve problems, provide fresh ideas, remove roadblocks and add direction and insight to your projects.8 They offer expert molecular design capabilities and use cutting-edge technologies to provide support for numerous client projects, across a range of disciplines.
To find out how Cresset Discovery Services can help you reach your next milestone faster and more cost effectively, contact us for a free confidential discussion.