The old problem and the new paradigm
Classical small molecule drug paradigms generally involve intervention at a ligand protein binding site. The binding event itself either stimulates or blocks normal protein function. As this is usually an equilibrium, (with some notable exceptions) it is then dependent on ligand concentration and stoichiometry. Modern drug discovery is often dogged by an absolute requirement to achieve and maintain an effective dose, in the relevant cellular compartment, that counters physiological concentration of the target protein.
A relatively new approach has emerged over the last 15 years which avoids some of these issues and is consequently rapidly becoming mainstream as a new therapeutic paradigm1-11. This approach focuses on effectively removing the target protein - to achieve the desired therapeutic outcome. This is done by subverting the natural protein degradation machinery. It is catalytic, not stochiometric, and is applicable to higher complexity / non-typical protein targets.
Protein degradation
Protein degradation is a fundamental component of cellular function. As part of the natural cycle; genes are transcribed, translated into proteins, subsequently folded, transported, modulated and then ultimately degraded and recycled. In most cells, proteins are tagged (poly-ubiquitinated) before being carried to the cells waste disposal unit ‘the proteasome’ which contains highly active peptide cleaving enzymes that break the protein down.
The tagging process itself is highly regulated: by (1) ubiquitin ligase enzymes E1, E2, E3 which coordinate to add single, or multiple, tags, and (2) their counter proteins - the ubiquitin specific proteases which cleave them back off (i.e., for de-tagging). Both protease and ligase systems are receiving huge R&D attention in academic institutions, pharmaceutical and biotech companies across the globe.
How the system can be worked with chemistry
The medicinal chemistry intervention most often being applied currently is to generate bifunctional ligands. In one component there is a target engaging ligand with a solvent accessible linking chemistry; in the second component an E3 ligase engaging ligand with its own solvent accessible link. The third component is a molecular string connecting the two through the linking functionality. The construct acts like a molecular glue that brings the target into proximity of the E3 ligase, in a similar way to normal substrate engagement, and ‘ideally’ leads to productive ubiquitination of specific surface lysine residues. Further ubiquitination initiates the degradation pathway for the target protein and the cycle can repeat. The current art is in (1) designing good binding ability for both components and (2) picking suitable linking trajectories and chemistry and of course (3) having the biology in place to assay ‘actual’ cellular degradation (figure 1).
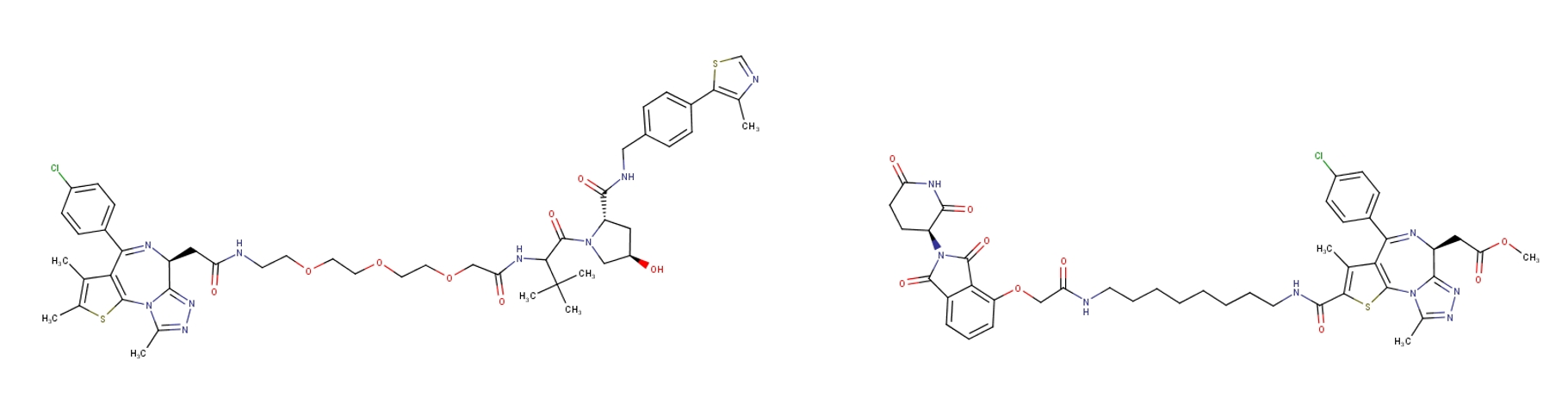
Figure 1: Two ‘BRD4 degrading’ bifunctional ligands; both containing a BRD4 binding component, a VHL binding tether (left) and Cereblon binding tether (right).
What can we do in terms of modeling?
A 3D molecular understanding of the complex binding events being used by these systems is essential in the design of these active therapeutics. Structural insights from modeling and X-ray crystallography have been critical. Small molecule ligand design and expansion into bifunctional ligands is a key facet of this approach which is of huge potential interest by many groups.
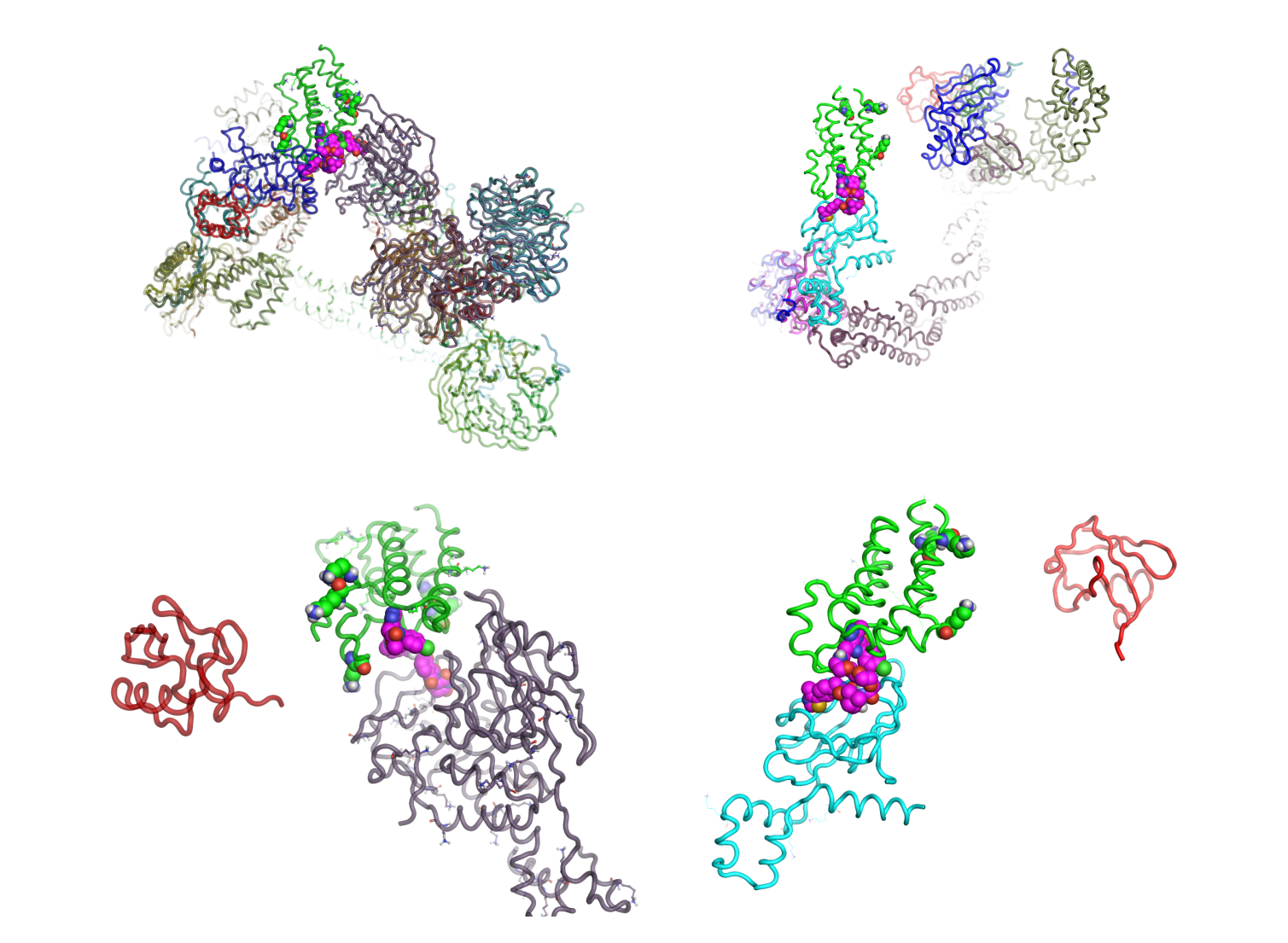
Figure 2: Top: Composite superimposition of Cereblon E3 ligase (left) and BRD4 in green, and VHL E3 ligase and BRD4 in green - modeled using Flare™. Complexes show the respective bifunctional ligands (magenta) in full context for ubiquitination and include PDB’s: 5T35, 5N4W, 4P5O, 2HYE and 6BN7. Bottom: Detail showing just the respective ubiquitins (in red) with both E3 ligase ends interacting via the bifunctional ligands with BRD4.
Figure 2 shows how much specific E3 ligases differ in their engagement with substrates. In this case the same target: BRD4 is manipulated through the different bifunctional ligands to undergo ubiquitination by conjugation through very different lysine residues. E2 ligase (in blue) holds ubiquitin (in red) in close enough proximity for transfer to BRD4 lysines.
Conclusion
Rational design of ligands without the random element currently necessary for linker design is feasible with some understanding of the higher order complexes that are operating. Additionally, Spark™ is an ideal platform in which to model linking variations or scaffold merging possibilities.
Apply Cresset expertise to your project
Contact us for a free confidential discussion to see how Cresset Discovery Services expert computational chemists can help advance your projects.
References
- Targeted Intracellular Protein Degradation Induced by a Small Molecule: En Route to Chemical Proteomics, Smitha A. R., Pucheaultb M., Taeb H. S. and Crews C. M, Bioorg Med Chem Lett. 2008 November 15; 18(22): 5904–5908.
- Protacs: Chimeric molecules that target proteins to the Skp1–Cullin–F box complex for ubiquitination and degradation, Sakamoto K, M., Kim K. B., Kumagai A., Mercurio F., Crews C. M, and Deshaies R. J., PNAS, 2001, vol. 98, no. 15, 8554.
- Selective Small Molecule Induced Degradation of the BET Bromodomain Protein BRD4, Zengerle M., Chan K. and Ciulli A., ACS Chem. Biol. 2015, 10, 1770−1777.
- Catalytic in vivo protein knockdown by small-molecule PROTACs, Nat Chem Biol. 2015 August; 11(8): 611–617, Bondeson et al.
- HaloPROTACS: Use of Small Molecule PROTACs to Induce Degradation of HaloTag Fusion Proteins, Buckley et al, ACS Chem Biol. 2015 August 21; 10(8): 1831–1837.
- Selective Target Protein Degradation via Phthalimide Conjugation, Winter G. E., Buckley D. L., Paulk J., Roberts J. M., Souza A., Dhe-Paganon S. and Bradner J. E., Science. 2015 June 19; 348(6241): 1376–1381.
- Development of protein degradation inducers of oncogenic BCR-ABL protein by conjugation of ABL kinase inhibitors and IAP ligands, Shibata N., Miyamoto N., Nagai K., Shimokawa K., Sameshima T., Ohoka N., Hattori T.,Imaeda Y., Nara H., Cho N. and Naito M., Cancer Sci 108 (2017) 1657–1666.
- Lessons in PROTAC Design from Selective Degradation with a Promiscuous Warhead, Bondeson D. P., Smith B. E., Burslem, G. M., Hamman B. D., Ishchenko A., Crews C. M., Cell Chemical Biology 25, 78–87, January 18, 2018.
- Next-Generation Drugs and Probes for Chromatin Biology: From Targeted Protein Degradation to Phase Separation, Cermakova K. and Hodges H. C. Molecules 2018, 23, 1958.
-
Small-molecule PROTACs: An emerging and promising approach for the development of targeted therapy drugs, An S., Fu L., EBioMedicine, 36 (2018) 553–562.
-
The PROTAC technology in drug development, Zou Y. Ma D. | Wang Y., Cell Biochem Funct. 2019, 37:21–30.

