Improving PROTAC properties via single-point changes to linkers
We explore how computational methods can be applied to proteolysis targeting chimera (PROTAC) design, to effectively tackle some of the ...
News
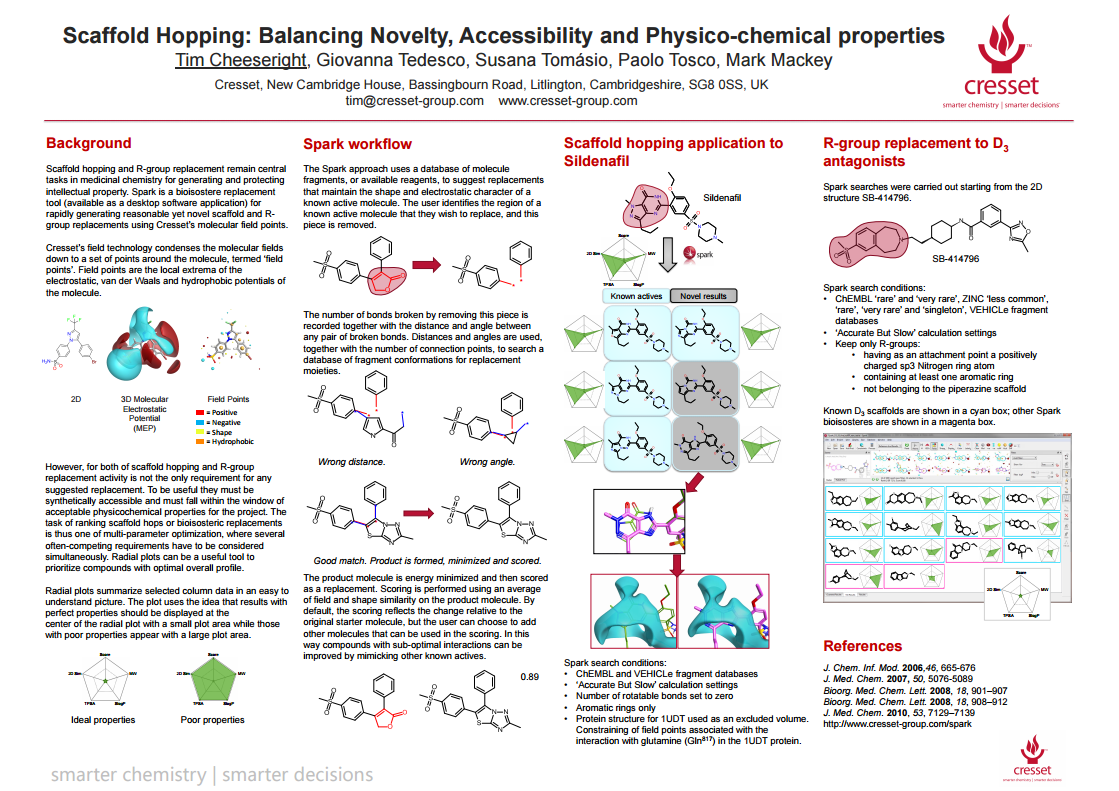
Scaffold hopping remains a central task in medicinal chemistry for generating and protecting intellectual property. We have previously presented a technique for rapidly generating reasonable yet novel scaffold replacements using molecular fields which has been extended to include R-group replacement. The approach uses a database of molecule fragments or available reagents to suggest replacements that maintain the shape and electrostatic character of a known active molecule.
However, for both of scaffold hopping and R-group replacement activity is not the only requirement for any suggested replacement. To be useful they must be synthetically accessible and must fall within the window of acceptable physicochemical properties for the project. The task of ranking scaffold hops or bioisosteric replacements is thus one of multi-parameter optimization, where several often-competing requirements have to be considered simultaneously.
In this poster we suggest methods to address these issues with reference to case studies of both scaffold hops and R-group replacements. Synthetic accessibility can be handled by tying the bioisostere search to addressable chemistry space, utilizing the chemist’s knowledge of what synthetic routes are feasible to guide the search. The best guide of novelty in a scaffold-hopping situation is the experience of the user: what other scaffold are known in the literature or in patents? To this end, we suggest a clear and minimal user interface to allow rapid triage of large result lists. Finally, assessing results in the light of physicochemical and predicted ADMET requirements can be achieved through a configurable radar plot giving clear visual feedback on how close any suggested replacement is to the ideal.
Click image to see poster.